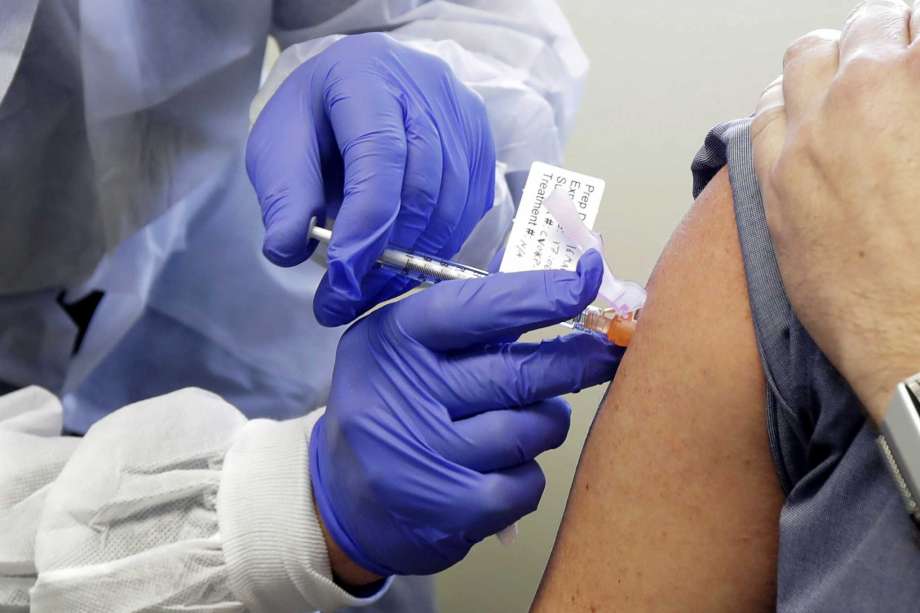
Serum Institute of India (SII), which has partnered with AstraZeneca for manufacturing the Oxford vaccine candidate for COVID-19, has sought permission from the Drugs Controller General of India (DCGI) for conducting phase 2 and 3 human clinical trials of the potential vaccine, highly-placed sources said on Saturday.
The sources told PTI that the Pune-based drug rm submitted its application to the DCGI on Friday seeking permission for conducting the trials of the vaccine candidate ‘Covishield’.
According to the application, it would conduct an observer-blind, randomised controlled study to determine the safety and immunogenicity of ‘Covishield’ in healthy Indian adults. The rm said that an around 1,600 participants of more than 18 years would be enrolled in the study,” a source said.
Initial results of the rst two-phase trials of the vaccine conducted in ve trial sites in the UK showed it has an acceptable safety prole and homologous boosting increased antibody responses, the source said.
India will ensure regulatory facilitation and actively consider supplementing resources if needed for expediting the Serum Institute of India’s phase 3 trial of a vaccine for COVID-19 developed by Oxford
University.
With the trials of COVID-19 vaccine being developed by AstraZeneca and Oxford University showing encouraging results, Serum Institute of India (SII) on Monday had said it will apply for licence from the Indian regulator to start clinical trials of the shot in a week’s time.
SII, the largest vaccine manufacturer in the world, has been chosen by Oxford and its partner AstraZeneca to manufacture the vaccine once it gets ready.
To introduce the vaccine, SII, the world’s largest vaccine maker by number of doses produced and sold, has signed an agreement to manufacture the potential vaccine developed by the Jenner Institute (Oxford University) in collaboration with British-Swedish pharma company AstraZeneca.
On the partnership with AstraZeneca, SII CEO Adar Poonawalla had said, “Serum Institute of India has entered a manufacturing partnership with AstraZeneca to produce and supply 1 billion doses of the COVID-19 vaccine being developed by Oxford University.”
These vaccines will be for India and middle and low income countries across the world (GAVI countries), he had said.
The rm plans to start the phase 2 and 3 human trials in India in August.
Oxford University on Monday announced satisfactory progress with the vaccine, making it one of the leading ones among the dozens of vaccine candidates being developed around the world.
According to a report in the Lancet, the initial trial results showed that the vaccine is safe and prompts protective immune response.
Last Monday, NITI Aayog member Dr V K Paul had said that India will ensure regulatory facilitation and actively consider supplementing resources if needed for expediting SII’s phase 3 trial of the vaccine developed by Oxford University.
“Regulatory facilitation will be ensured. It is already being undertaken and being done. If resources need to be supplemented, that will be actively considered.
“The government will leave no stone unturned to ensure that the people of India and the international community has access to an Indian vaccine as early as possible,” Paul had said.

























 WhatsApp us
WhatsApp us
Pingback: 토렌트 사이트
Pingback: Online medicatie kopen zonder recept bij het beste Benu apotheek alternatief in Amsterdam Rotterdam Utrecht Den Haag Eindhoven Groningen Tilburg Almere Breda Nijmegen Noord-Holland Zuid-Holland
Pingback: ดูซีรี่ย์ออนไลน์